Electrolysis III – Faraday’s Laws of Electrolysis SS2 Chemistry Lesson Note
Download Lesson NoteTopic: Electrolysis III – Faraday’s Laws of Electrolysis
FARADAY’S LAWS OF ELECTROLYSIS
FARADAY’S FIRST LAW OF ELECTROLYSIS
Faraday’s first law states that the mass (m) of an element discharged during the electrolysis of an electrolyte is directly proportional to the quantity of electricity (Q) passing through it.
Faraday’s first law can be expressed in the following mathematical way:
= Mis directly proportional to Q
But Q = It
:. M is directly proportional to lt
M = Elt or
M = EQ
Where E is the constant of proportionality known as the Electrochemical equivalent of a substance.
M is measured in grams (g)
I is measured in Amperes (A)
t is measured in seconds (s)
Q is measured in Coulomb(C)
Therefore E is measured in gA^{-1}S^{-1} or gC^{-1} for a particular electrolytic process at different times, the law can also be expressed as M_{1}/Q_{1}=M_{2}/Q_{2}
OR: M_{1}=M_{2}
I_{1} t_{1}
I_{2}
VERIFICATION OF FARADAY’S FIRST LAW OF ELECTROLYSIS.
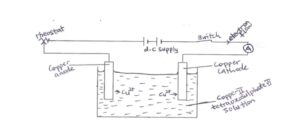
The experimental setup is
The electrolytic cell above can be used to verify Faraday’s first law.
The copper cathode is weighed at the beginning of the experiment. A steady current of 1A is passed for 10 mins and the current supply is switched off. The copper cathode is then weighed again to find the gain in mass. This procedure is repeated using different time intervals. A graph of gain in mass against time gives a straight line passing through the origin.
The copper cathode is then weighed again to find the gain in mass. This procedure is repeated using different time intervals. A graph of gain in mass against time gives a straight line passing through the origin.
When the experiment is repeated using different currents for a fixed period, a graph of gain in mass against the magnitude of the current also gives a straight line passing through the origin.

The two graphs show that the mass of copper deposited is directly proportional to the magnitude of the current, i.e M is directly proportional to l
FARADAY’S SECOND LAW OF ELECTROLYSIS
Faraday’s second law of electrolysis states that when the same quantity of electricity Q is passed through different electrolytes, the relative number of moles of the elements discharged is inversely proportional to the charges on the ions of the elements.

The minimum quantity of electricity required to liberate one mole of a univalent ion is 96500C. This quantity of electricity is called the Faraday (F). One Faraday (1F) is one mole of electrons which contains Avogadro’s number (6.62\times 10^{23}) of electrons. It follows that two Faraday’s (2F) will be required to discharge 1 mole of ions of a divalent element or radical such as Pb^{2+} or SO_{4}^{2-}
i.e M^{2+}+2e- \underline{A}^{-} A + M or 2e-
VERIFICATION OF FARADAY’S SECOND LAW OF ELECTROLYSIS
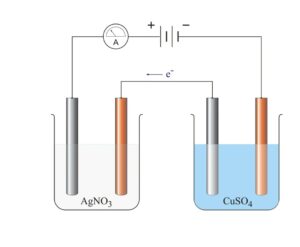
To verify the second law, set up two electrolytic cells.
I. One with copper electrodes immersed in CuSO4 solution
ii. The other with silver electrodes immersed in AgNO3 solution.
iii. The copper and silver cathodes are weighed before the experiment begins.
A steady current of 1A is passed into the solution for say 15 minutes and the current supply is switched off. The mass of each cathode is then weighed again to find the gain in mass. The number of moles of silver deposited at the silver cathode will be doubled the number of copper deposited at the copper cathode although the same quantity of electricity was passed through the two electrolytes i.e. if Y moles of copper are deposited by 2Y moles of silver are deposited. The charge on the Cu2+ is +2 and that on the Ag+ is +1 hence, the number of moles of copper and silver deposited is inversely proportional to the charge on their ions
CALCULATIONS BASED ON THE LAW
Examples:
- Calculate the electrochemical equivalent of a metal M if a current of 5A is passed for 1hr and 2g of M is deposited
Solution:
:- M = 2g, 1 = 5A,
M = Z It
t = 1hr
Z X 5 x 1 x 60 x 60
Z=2.78 x 104 g 11 s¹
APPLICATIONS OF ELECTROLYSIS
Electrolysis is applied in the industries in the following areas e.g.
- Extraction of elements: Na, K, Mg, Ca, Al, Zn and nonmetals e.g. H2, F2, and Cl2 are obtained either by electrolysis of their ores or by the electrolysis of their fused compounds or their aqueous solutions.
- Purification of metals e.g. Cu, Hg, Ag, Au.
- Electroplating of one metal by another.
- Preparation of certain important compounds such as sodium hydroxide, and sodium trioxochlorate (V)
ELECTROPLATING
Electroplating is a method of coating the surface of one metal with another metal, usually copper, silver, chromium, nickel, or gold using electrolysis. The main objectives of electroplating are:
- Decoration
- Protection against corrosion.
Articles which are usually plated include cutlery, jewellery and iron or steel objects such as the metallic parts of motor cars.